Opdateret: 6. marts 2026
Fluoguide AS (FLUO.ST) kursmål 2026: 274% upside – Aktieanalyse
33,20
Fluoguide AS kursmål 2026: Analytikere ser 274% upside
Det gennemsnitlige kursmål for Fluoguide AS er 124.22 Kr.. Dette kursmål repræsenterer analytikernes vurdering af aktiens pris de næste 12 måneder. Aktien er muligvis undervurderet, da kursmålet er højere end den nuværende kurs. Aktien er undervurderet med 274,16%, hvilket kan indikere et potentielt afkast inden for de næste 12 måneder, hvis analytikernes vurdering holder. Vurder også aktiens nøgletal som P/E, P/S og P/B samt den tekniske analyse længere nede på siden for et komplet billede.Er Fluoguide AS overvurderet eller undervurderet?
Fluoguide AS er i øjeblikket undervurderet med en afvigelse på 274.2% i forhold til kursmålet.
Den aktuelle kurs er Kr.33.2, hvilket placerer Fluoguide AS i kategorien undervurderet. Aktien vurderes som undervurderet, hvis kursen er under Kr.111.798, og som overvurderet, hvis kursen overstiger Kr.136.642.
Fair value-intervallet er defineret som ±10% af kursmålet, da kursmål ofte er skøn og ikke eksakte værdier. For Fluoguide AS ligger fair value-intervallet mellem Kr.111.798 og Kr.136.642.
Sidder du med Fluoguide AS i porteføljen? Så læs det her.
Fluoguide AS scorer kun 14 ud af 100 i vores analyse. Aktien er i nedtrend, og mange investorer sidder fast i en position der taber penge. Stop med at gætte – få en gratis porteføljeanalyse og find ud af, om dine aktier trækker din portefølje ned. Vi har hundredvis af aktier der scorer langt højere.
Få gratis porteføljeanalyse →Om Fluoguide AS – Bioteknologi
FluoGuide A/S er et life science-selskab, der udvikler løsninger til kirurgi. Helt basalt hjælper de kirurger med at se kræftvæv under operationen. Deres hovedprodukt er FG001, som de tester i fase I/II mod aggressiv hjernekræft og i fase II mod lungekræft. FluoGuide arbejder også på FG002, en teknologi, der bruger lys til at skelne kræft fra normalt væv. Virksomheden driver udviklingen fra København, hvor de har base. De startede i 2018 og samarbejder blandt andet med Linköping Universitetshospital om FG001. FluoGuide satser på at levere bedre præcision i kræftkirurgi. Læs om virksomhedenNøgletal for Fluoguide AS
Hvad er Fluoguide AS for en type aktie?
Fluoguide AS klassificeres som en spekulativ aktie.
Score-oversigt for Fluoguide AS
📈 Værdiansættelse
P/S-ratioen er 2,474.7 (høj — markedet betaler en premium for omsætningen). P/B er 7.1.
🏦 Finansiel Sundhed
Piotroski-scoren er 3 ud af 9 — svag finansiel sundhed, vær opmærksom. Altman Z-score er 10.11 (sikker zone — lav konkursrisiko). Gældsgraden (D/E) er 23.0% — lav gearing, solid balance.
Hvornår har Fluoguide AS regnskab – Næste dato: 26/05-2026
Fluoguide AS's næste regnskab bliver offentliggjort d. 26. maj 2026. Dette regnskab dækker første kvartal (Q1) og sluttede d. 31. marts 2026.Teknisk analyse af Fluoguide AS 2026 – Signal: Stærkt Sælg
Baseret på trendanalyse (SMA50/SMA200) og MACD-trend (positiv/negativ på daglig + ugentlig basis). Opdateret 07. marts 2026.
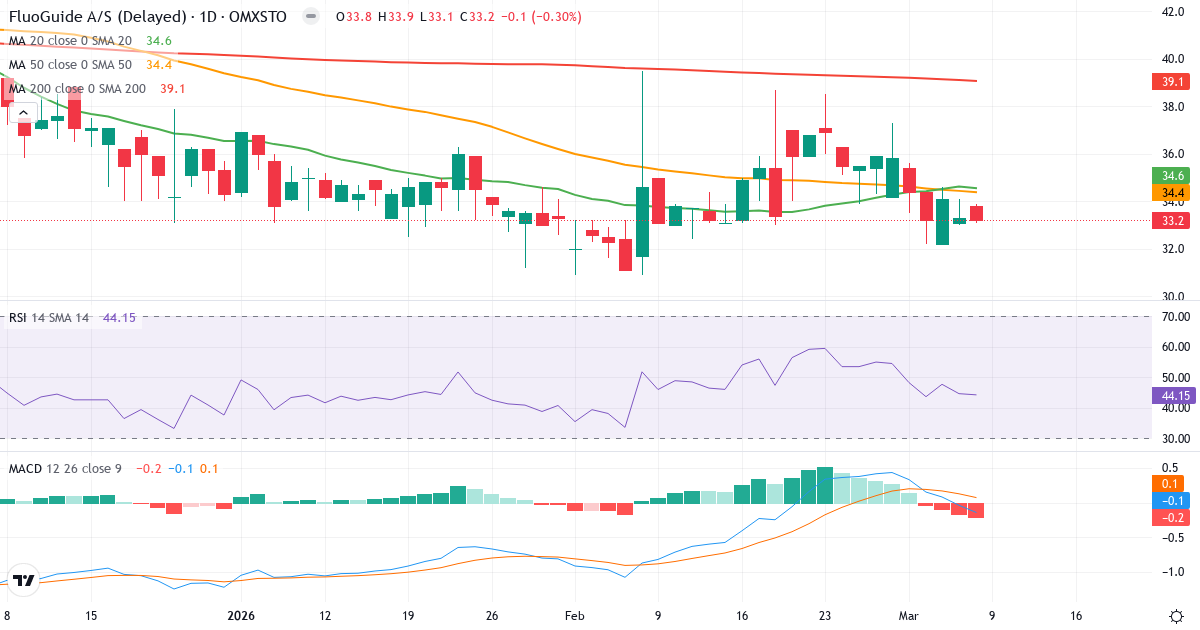
Trendanalyse af Fluoguide AS
Fluoguide AS er i en kortsigtet nedtrend. Kursen på 33.20 Kr. ligger 3.9% under 20-dages gennemsnittet (34.56 Kr.). På mellemlang sigt er billedet negativt: kursen ligger 3.4% under 50-dages gennemsnittet (34.38 Kr.). Langsigtet ligger aktien 15.0% under 200-dages gennemsnittet (39.06 Kr.), hvilket indikerer en langsigtet nedtrend.
Death Cross: 50-dages gennemsnittet (34.38) ligger under 200-dages (39.06), hvilket er et klassisk bearish signal for Fluoguide AS.
Er Fluoguide AS overkøbt eller oversolgt?
Fluoguide AS har en RSI på 44.2. Aktien befinder sig i den lave del af neutral zone, hvilket tyder på svagt momentum uden at være oversolgt. I den nuværende nedtrend bør man være forsigtig med at tolke dette som en købsmulighed.
MACD-analyse for Fluoguide AS
Daglig MACD: MACD er negativ (-0.141), hvilket betyder at den kortsigtede trend er bearish (12-dages EMA ligger under 26-dages EMA). MACD ligger under signallinjen (0.073) med 0.214, hvilket bekræfter tiltagende negativt momentum i nedtrenden.
Ugentlig MACD: MACD5 er negativ (-1.777), hvilket indikerer en overordnet bearish langsigtet trend. MACD5 ligger under signallinjen (-1.610) med 0.167, hvilket bekræfter vedvarende negativt langsigtet momentum.
Samlet MACD-vurdering: Både daglig og ugentlig MACD er negative og under deres signallinjer. Trend og momentum er negativt på tværs af tidshorisonter – det stærkeste bearish signal.
Risikoprofil for Fluoguide AS
Fluoguide AS har en årlig standardafvigelse på 49.2%, hvilket klassificerer aktien som høj risiko. Betydelige kursudsving er normalt, og aktien kræver en højere risikotolerance.
Samlet teknisk konklusion for Fluoguide AS
Trend: Negativ. Kursen handler under SMA50 og SMA200.
MACD Trend (nullinje): Daglig bearish (-0.141). Ugentlig bearish (-1.777).
MACD Momentum (signal): Daglig faldende. Ugentlig faldende.
RSI: 44.2 – neutral.
Teknisk signal: Stærkt Sælg
Kursen er i nedtrend (under SMA50/SMA200) og MACD er negativ på både daglig og ugentlig basis. Undgå køb og overvej at lukke eksisterende positioner.