Saniona (SANION.ST) kursmål 2026/2027: 55% potentiale – anbefaling og Aktieanalyse
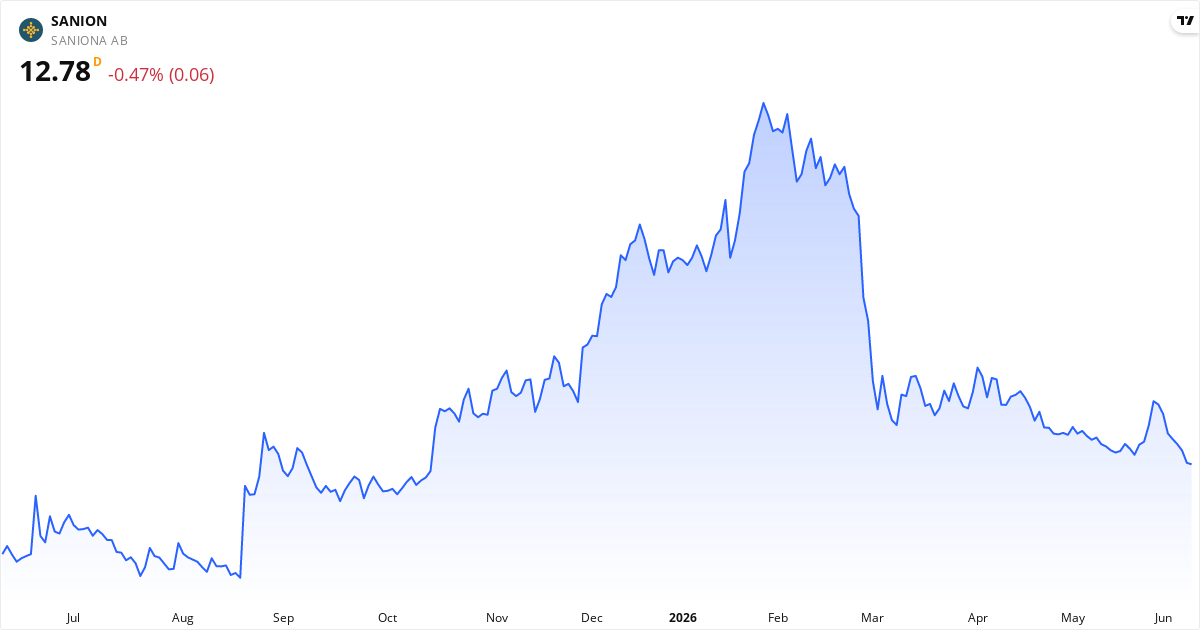
Saniona kursmål 2026: Analytikere ser 54% potentiale
Det gennemsnitlige kursmål for Saniona er 20.00 Kr.. Dette kursmål repræsenterer analytikernes vurdering af aktiens pris de næste 12 måneder.Forskellen mellem kursmålet og den nuværende kurs kaldes potentialet (på engelsk "upside"). Det viser, hvor meget analytikerne forventer aktien kan stige op til kursmålet over de næste 12 måneder. Hvis kursen omvendt allerede ligger over kursmålet, mener analytikerne aktien er for dyr — og forventer at aktien skal falde.
Aktien er undervurderet med 53,85% i forhold til analytikernes kursmål. Hvis vurderingen holder, kan det indikere et potentielt afkast inden for de næste 12 måneder. Vurder også aktiens nøgletal som P/E, P/S og P/B samt den tekniske analyse længere nede på siden for et komplet billede. Hvad betyder det for dig? Kursmålet er analytikernes bedste bud – ikke en garanti. Tjek også virksomhedens tal, kursudviklingen og hvor stor risiko du kan tåle, før du beslutter dig.
Er Saniona overvurderet eller undervurderet?
Saniona er i øjeblikket undervurderet med en afvigelse på 53.8% i forhold til kursmålet.
Den aktuelle kurs er 13,00 Kr, hvilket placerer Saniona i kategorien undervurderet. Aktien vurderes som undervurderet, hvis kursen er under 18,00 Kr, og som overvurderet, hvis kursen overstiger 22,00 Kr.
Fair value-intervallet – det vi her kalder et rimeligt prisleje for aktien – er defineret som ±10% af analytikernes kursmål, da et kursmål altid er et skøn og ikke en eksakt værdi. For Saniona ligger det rimelige prisleje mellem 18,00 Kr og 22,00 Kr.
Om Saniona – Bioteknologi
Saniona er et dansk firma, der udvikler medicin, især til sygdomme i hjernen og spiseforstyrrelser. Virksomheden blev grundlagt i 2011 og har hovedkvarter i Ballerup. De driver forskning i både tidlige og sene faser. Saniona er især kendt for Tesofensine, som er i fase III kliniske forsøg til behandling af fedme. De laver også Tesomet, som de tester mod Prader-Willi syndrom. Derudover udvikler Saniona NS2359, der skal hjælpe folk med kokainafhængighed. De har altså en bred vifte af lægemiddelkandidater. De fokuserer på at få deres produkter gennem de kliniske forsøg, så de kan komme på markedet. Læs om virksomhedenSaniona hører til temaet Biotekaktier. Se alle biotekaktier rangeret efter signal og momentum →
Nøgletal for Saniona
Nøgletal hjælper dig med hurtigt at vurdere, om aktien er billig eller dyr. P/E viser hvor mange års nuværende indtjening du betaler for aktien – lavere er typisk billigere. P/S og P/B sammenligner kursen med selskabets omsætning og bogførte værdi. Tallene siger mest, når man sammenligner med andre selskaber i samme branche.
Hvad er Saniona for en type aktie?
Saniona klassificeres som en vækstaktie og value-aktie. Det betyder at aktien kombinerer flere attraktive egenskaber.
Score-oversigt for Saniona
Saniona ser billig ud i forhold til de nøgletal vi måler på. Det kan være positivt, hvis virksomheden samtidig har god kvalitet og en stabil kursudvikling.
Virksomheden ser blandet ud. Der er nogle styrker, men også ting man bør holde øje med.
Saniona har svag kursudvikling. Markedet viser lige nu ikke stor interesse for aktien.
Samlet set ser Saniona blandet ud. Der er både positive og negative forhold.
📈 Værdiansættelse
Er aktien billig eller dyr? Disse nøgletal sammenligner kursen med selskabets indtjening, omsætning og bogførte værdi.
Saniona handles til en P/E på 7.9, hvilket er under markedsgennemsnittet og kan indikere en attraktiv pris. P/S-ratioen er 4.3 (i moderat niveau). P/B er 3.0 (moderat niveau).
💵 Rentabilitet & Vækst
Tjener selskabet penge? Disse tal viser hvor god virksomheden er til at omdanne omsætning og kapital til overskud.
Saniona har en overskudsgrad (hvor stor del af omsætningen der bliver til overskud) på 51.1% — fremragende. ROE (afkast på egenkapital — hvor godt selskabet udnytter aktionærernes penge) er 52.2% — meget højt. ROA (afkast på alle aktiver — hvor godt selskabet udnytter alt det det ejer) er 30.1% — meget højt. Den seneste kvartalsindtjening voksede med 455.7% år-over-år — stærk vækst.
🏦 Finansiel Sundhed
Er selskabet finansielt sundt? Disse mål vurderer balance, gæld og risikoen for økonomiske problemer.
Piotroski-scoren (et mål for finansiel sundhed på en skala fra 0 til 9, hvor højere er bedre) er 3 ud af 9 — svag finansiel sundhed, vær opmærksom. Altman Z-score er 22.14 (sikker zone — lav konkursrisiko). Gældsgraden (D/E) – forholdet mellem gæld og egenkapital – er 46.6% – lav gældsætning og en solid balance.
🚀 Momentum & Short Interest
Hvad gør kursen lige nu? Viser kursens udvikling over forskellige tidsperioder og hvor mange investorer der satser på fald.
Short interest er 0.71% — lavt, markedet forventer ikke et fald.
Hvornår har Saniona regnskab?
Saniona aflagde senest regnskab for 1532 dage siden (d. 31. marts 2022, Q1 2022) – og skuffede ift. analytikernes forventninger. Den næste regnskabsdato er endnu ikke offentliggjort – tjek virksomhedens finansielle kalender for opdateringer. Se beat/miss-sektionen nedenfor for detaljer på det seneste regnskab.Slog Saniona analytikernes forventninger?
Hvorfor er det vigtigt? Når et selskab offentliggør regnskab, sammenligner markedet det med analytikernes forventninger på forhånd. Hvis selskabet slår forventningerne (beat), stiger kursen typisk. Hvis selskabet skuffer (miss), falder den ofte. Et selskab der gang på gang slår forventningerne, viser sig som en stabil leverer – det modsatte er en advarselslampe.
Saniona har skuffet i 5 ud af de seneste 5 kvartaler – det er flere skuffelser end positive overraskelser. Det kan tyde på, at analytikerne er for optimistiske, eller at selskabet har svært ved at indfri forventningerne.
Vi viser kun kvartaler, hvor analytikerne på forhånd havde et offentligt EPS-estimat. EPS (Earnings Per Share) betyder hvor meget selskabet tjente pr. aktie i kvartalet. "Slog forventningerne" = mindst 2% bedre end estimatet, "Skuffede" = mindst 2% dårligere. Hvis EPS-estimatet er meget lille, vises store procent-afvigelser som "langt over/under forventet" frem for et misvisende tal.
Teknisk analyse af Saniona 2026 – Signal: Stærkt Sælg
Baseret på trendanalyse (SMA50/SMA200) og MACD-trend (positiv/negativ på daglig + ugentlig basis). Opdateret 10. juni 2026.
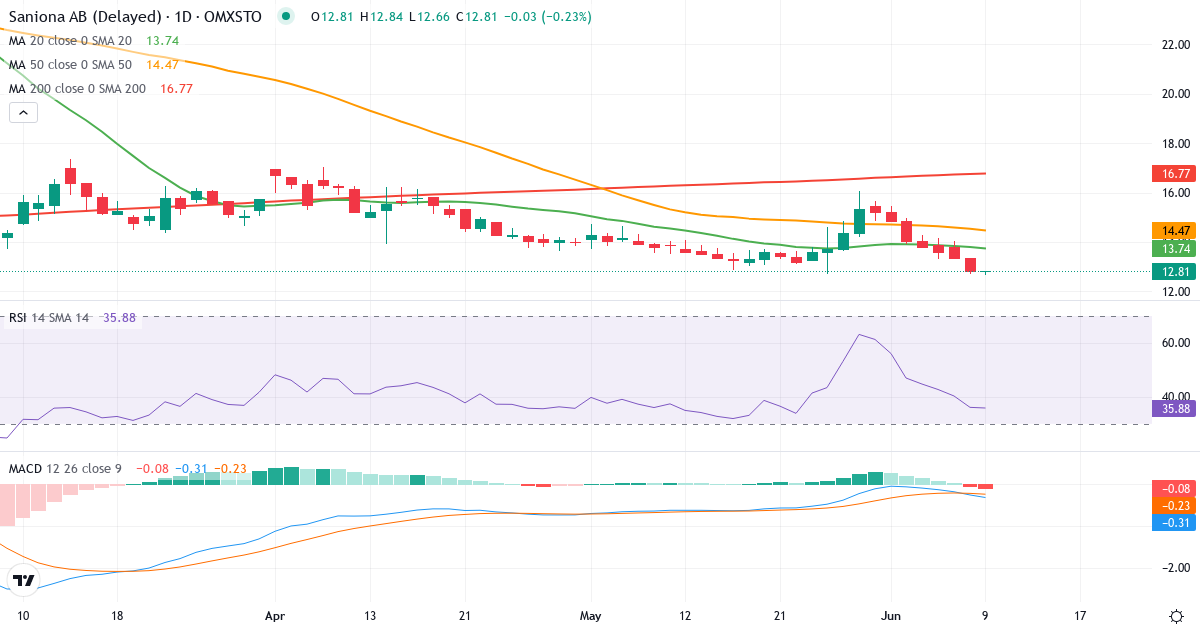
Trendanalyse af Saniona
Saniona er i en kortsigtet nedtrend. Kursen på 13.00 Kr. ligger 5.4% under 20-dages gennemsnittet (13.75 Kr.). På mellemlang sigt er billedet negativt: kursen ligger 10.2% under 50-dages gennemsnittet (14.47 Kr.). Langsigtet ligger aktien 22.5% under 200-dages gennemsnittet (16.77 Kr.), hvilket indikerer en langsigtet nedtrend.
Death Cross: 50-dages gennemsnittet (14.47) ligger under 200-dages (16.77) – det er et klassisk negativt signal og tyder på, at den mellemlange trend i Saniona er svagere end den lange.
Er Saniona overkøbt eller oversolgt?
Saniona AB (publ) har en RSI på 37.9. Aktien ligger i den lave del af det neutrale område, hvilket tyder på svag kursudvikling uden at være decideret oversolgt. I den nuværende faldende trend bør man være forsigtig med at tolke dette som et købssignal.
MACD-analyse for Saniona
MACD er et signal, der sammenligner aktiens korte og lidt længere trend. Når MACD er over 0, peger den korte trend op. Når MACD ligger over sin signal-linje, har aktien medvind. Vi viser både den daglige MACD (kort sigt) og den ugentlige (lang sigt), så du kan se om begge tidshorisonter peger samme vej.
Daglig MACD: MACD er negativ (-0.300), hvilket betyder at den korte trend peger ned – sælgerne har overtaget på kort sigt. MACD ligger under signal-linjen (-0.232) med 0.068, hvilket bekræfter at aktien har modvind – nedturen styrkes.
Ugentlig MACD: Den ugentlige MACD er negativ (-0.920), hvilket betyder at den længere trend peger ned. Den ligger under signal-linjen (-0.153) med 0.767, hvilket bekræfter vedvarende langsigtet modvind.
Samlet MACD-vurdering: Både den daglige og ugentlige MACD er negative og under deres signal-linjer. Trenden peger ned både på kort og lang sigt – det stærkest mulige negative signal.
Risikoprofil for Saniona
Standardafvigelse er et mål for, hvor meget aktiens kurs typisk svinger på et år. Højere tal betyder større kursudsving og dermed højere risiko.
Saniona AB (publ) har en årlig standardafvigelse på 84.1%, hvilket klassificerer aktien som ekstrem risiko. Kun risikovillige investorer bør overveje denne aktie, da kursudsving kan være meget store.
Samlet teknisk konklusion for Saniona
Overordnet trend: Faldende. Kursen ligger under sit 50-dages gennemsnit og sit 200-dages gennemsnit – det bekræfter en svag trend.
Trend (MACD): Kort sigt: peger ned (daglig værdi -0.300); Lang sigt: peger ned (ugentlig værdi -0.920).
Styrke (MACD vs. signal-linje): Kort sigt: aktien har modvind; Lang sigt: modvind.
RSI: 37.9 – neutral (kursudviklingen er hverken voldsomt presset op eller ned).
Teknisk signal: Stærkt Sælg
Kursen ligger under både 50- og 200-dages gennemsnit, og MACD er negativ på både kort og lang sigt. Undgå køb og overvej at lukke eksisterende positioner.
Ofte stillede spørgsmål om Saniona aktien i 2026
Skal man købe Saniona aktien nu?
Hvad er kursmålet for Saniona aktien?
Er Saniona en udbytteaktie?
Hvad er risikoen i Saniona aktien?
Er Saniona overvurderet — er den blevet for dyr?
Er Saniona overkøbt?
Hvornår har Saniona næste regnskab?
Er Saniona shortet?
Køber insidere (ledende medarbejdere og direktøren) aktier i Saniona?
Er Saniona en god aktie?
Hvad er fremtiden for Saniona aktien?
Hvorfor bevæger Saniona aktien?
Hvor meget kan Saniona stige?
Hvor lavt kan Saniona falde?
Hvad laver Saniona?
Har Saniona opjusteret eller nedjusteret forventningerne?
Tjener Saniona penge eller giver underskud?
Kan Saniona gå konkurs?
Har Saniona meget gæld?
Seneste nyheder om Saniona-aktien 2026
- Dato: 2026-06-04 08:00:00
Kategori: Øvrige ændringer
Nyheden: Saniona annoncerer et virtuelt R og D arrangement den 17. juni 2026 med fokus på SAN2668 mod svære pædiatriske epilepsier. Webinaret gennemgår sygdomsområde og behandlingslandskab, videnskabelig begrundelse, prækliniske data samt planlagt fase 1 strategi og kommercielt potentiale. Ekstern ekspert Nicola Specchio deltager, og der afsluttes med live spørgerunde. Nyheden giver primært øget indblik i pipeline uden nye finansielle nøgletal eller konkrete aftaler. (dato: 2026-06-04 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2026-05-27 17:15:00
Kategori: Øvrige ændringer
Nyheden: Generalforsamlingen godkendte årsrapporten for 2025 og besluttede ingen udbyttebetaling. Bestyrelsen blev genvalgt med John Haurum som bestyrelsesformand og Jørgen Drejer som næstformand, og PwC blev genvalgt som revisor. Aktionærerne vedtog også rammer for nomineringskomiteen samt vederlag til nomineringskomiteens eksterne medlemmer. Generalforsamlingen besluttede ikke at godkende vederlagsrapporten for 2025. Bestyrelsen trak desuden forslag om bemyndigelse til udstedelser samt forslag om et langsigtet incitamentsprogram tilbage, da større aktionærer forventedes at stemme imod, og selskabet vil i stedet arbejde på et nyt forslag til en senere generalforsamling (dato: 2026-05-27 17:15)
Læs mere her: Hent pdf med detaljer - Dato: 2026-05-26 08:00:00
Kategori: Regnskab
Nyheden: Saniona offentliggør Q1 2026 regnskab. Omsætning 4,6 mio. SEK mod 9,8 mio. SEK året før og driftsresultat -56,5 mio. SEK mod -16,5 mio. SEK. Nettoresultat -46,2 mio. SEK og EPS -0,33. Kontantbeholdning 532 mio. SEK pr. 31 marts 2026. Selskabet har færdiggjort GLP toksikologi materiale for tre interne programmer og forventer fase 1-start for SAN2668 og SAN2465 i Q4 2026 samt SAN2219 i 1H 2027. Partnerprogrammer kan give nærfristede milepæle, bl.a. 7,5 mio. USD ved Jazz fase 1-start af SAN2355 og 10 mio. USD ved Acadia fase 2-start af ACP-711. (dato: 2026-05-26 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2026-04-30 13:05:00
Kategori: Regnskab
Nyheden: Saniona offentliggør årsrapporten for 2025 på selskabets hjemmeside. Rapporten viser omsætning på 434,4 mio. SEK og et driftsresultat på 271,0 mio. SEK samt årets resultat på 284,7 mio. SEK. Likvidbeholdningen udgjorde 580,8 mio. SEK ved årets udgang. Året var præget af partneraftaler, især licensaftalen med Jazz for SAN2355 med upfront-betaling på 42,5 mio. USD, samt fremskridt i den interne pipeline mod planlagte fase 1-studier i 2026. Selve publiceringen af årsrapporten ændrer typisk ikke casen væsentligt, da hovedtal ofte er kendt fra tidligere meddelelser. (dato: 2026-04-30 13:05)
Læs mere her: Hent pdf med detaljer - Dato: 2026-04-24 17:35:00
Kategori: Øvrige ændringer
Nyheden: Saniona indkalder til ordinær generalforsamling 27 maj 2026 og fremlægger forslag, herunder ingen udbytte, genvalg af bestyrelse og revisor samt justeret bestyrelseshonorar. Bestyrelsen foreslår også bemyndigelse til at udstede nye aktier, tegningsretter og konvertibler op til ca. 20 procent af aktiekapitalen frem til næste generalforsamling. Derudover foreslås et langsigtet incitamentsprogram LTI 2026 med RSU på op til 1.347.300 aktier svarende til ca. 0,97 procent potentiel udvanding, inkl. teknisk set-up med serie C aktier til levering. (dato: 2026-04-24 17:35)
Læs mere her: Hent pdf med detaljer - Dato: 2026-02-26 07:00:00
Kategori: Regnskab
Nyheden: Saniona offentliggør årsrapport 2025: omsætning 434,4 mio. SEK (+30%), driftsresultat 271,0 mio. SEK og nettoresultat 284,6 mio. SEK (+51%). Q4 var svagt med omsætning 4,6 mio. SEK og underskud -41,7 mio. SEK pga. fravær af engangsindtægter. Kontantbeholdning 580,8 mio. SEK og egenkapitalandel 93 %. Året blev løftet af 404,8 mio. SEK i upfront fra Jazz og fremskridt i partnerskaber. Pipeline: to fase 1-studier planlagt i 2. halvår 2026 og et tredje primo 2027. Bestyrelsen foreslår intet udbytte. (dato: 2026-02-26 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2026-02-20 08:00:00
Kategori: Øvrige ændringer
Nyheden: Saniona inviterer investorer til Q4- og årsopkald den 26. februar 2026 kl. 11:00 CET. Ledelsen præsenterer resultater og en selskabsopdatering, efterfulgt af spørgsmål/svar. Optagelse lægges på saniona.com og redeye.se. Meddelelsen ændrer ikke selskabets forventninger og vurderes neutral for kursen, men kan give ny indsigt i projektportefølje og strategi. (dato: 2026-02-20 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2025-12-17 08:00:00
Kategori: Øvrige ændringer
Nyheden: Saniona meddeler flere ledelsesudnævnelser på tværs af klinisk udvikling, translational medicin, toksikologi og CMC. Formålet er at styrke eksekvering og fremskynde selskabets CNS pipeline: SAN2219 (refraktære fokale anfald), SAN2465 (depression) og SAN2668 (pædiatrisk epilepsi). Alle tre er finansieret frem til fase 1 læsninger, understøttet af nyligt indleverede patentansøgninger med beskyttelse ind i 2040erne, og fase 2 forventes start i 2028. De nye profiler inkluderer Head of Clinical Operations, Head of Clinical Development, Director of Translational Medicine, Head of Toxicology og Head of CMC. Nyheden forbedrer operationel kapacitet og kan sænke udviklingsrisiko, men indeholder ingen finansielle mål, data eller nye tidsmæssige katalysatorer. Kortsigtet vurderes kursdriver begrænset, langsigtet potentielt positiv for eksekvering. (dato: 2025-12-17 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2025-11-27 08:00:00
Kategori: Regnskab
Nyheden: Saniona leverer et stærkt Q3 2025 drevet af licensaftale med Jazz. Omsætning steg til SEK 410,7 mio fra 7,2 mio. Driftsresultat blev SEK 359,9 mio mod minus 18,9 mio. Nettoresultat SEK 329,6 mio og EPS 2,39. Kontanter SEK 672,8 mio. Selskabet modtog USD 42,5 mio i upfront for SAN2355 og kan modtage milepæle op til USD 992,5 mio samt royalty. Pipeline styrkes med udvælgelse af SAN2668. Resultatet forbedrer finansiel styrke og reducerer risiko. (dato: 2025-11-27 08:00)
Læs mere her: Hent pdf med detaljer - Dato: 2025-11-06 08:00:00
Kategori: Øvrige ændringer
Nyheden: Saniona offentliggør sammensætningen af nomineringsudvalget for generalforsamlingen 2026. Dette er en standardmeddelelse uden direkte indvirkning på aktien. (dato: 2025-11-06 08:00)
Læs mere her: Hent pdf med detaljer
